Emergency Use Authorization (EUA) KN95 Face Masks Explained | FDA Guidance on Personal Protection Equipment
Emergency Use Authorization from the FDA: what is it and what does it have to do with the current COVID-19 Coronavirus pandemic? With many businesses and school districts making progress to reopening, there’s likely going to be a renewed need for PPE in the weeks and months to come. To avoid shortages, the FDA is using Emergency Use Authorizations as a tool to help make more types of PPE available for those in need, but it’s important to understand what that entails and what that means about the PPE you can buy.
At the center of this conversation are KN95 face masks, one of the most effective pieces PPE against the coronavirus. However, how exactly does an EUA make it easier to get a KN95 mask? Here’s all you need to know.
 What Is a EUA?
What Is a EUA?
The Emergency Use Authorization is an ability that the FDA has to facilitate the availability and use of certain items during public health emergencies. While this has been used in the past, it’s particularly pertinent in the current COVID-19 crisis. To understand how it works, we first need to explain the difference between a KN95 mask and an N95 mask, the normal respirator masks you see in the U.S.
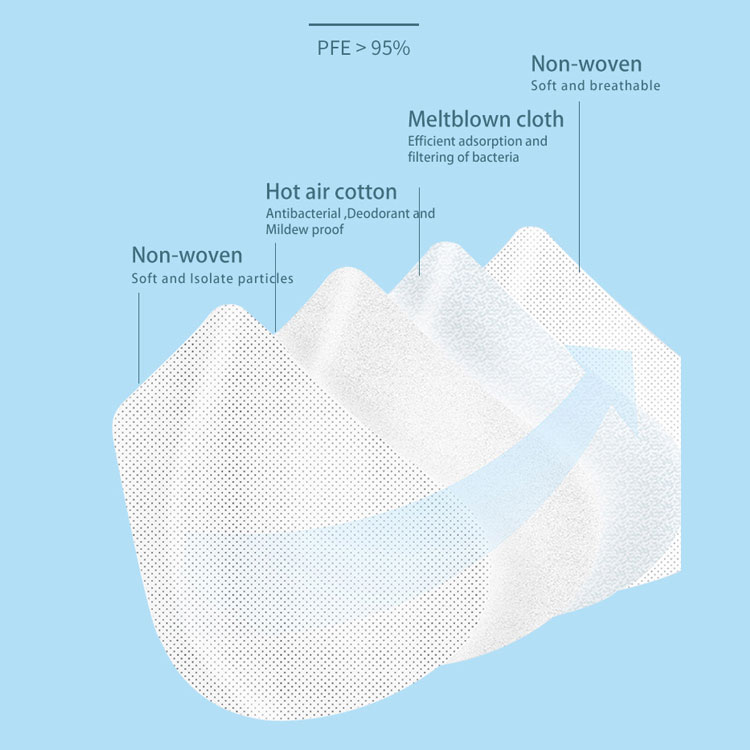
An N95 mask is made and approved in accordance with requirements established by the U.S. National Institute for Occupational Safety and Health (NIOSH). By comparison, KN95 masks are made by Chinese standards. With this said, the two are quite similar otherwise. Here’s a close look at some of the similarities between N95 and KN95 masks.
- Both filtration systems are tested by capturing salt particles
- Both are tested with a flow rate of 85L per minute and need to have a 95% standard filtration performance
- Both masks are able to block many particles smaller than 0.3 microns. That’s the testing size used for both sets of masks. For comparison, COVID-19 particles are less than 0.14 microns
At first glance, it seems like both sets of masks are almost identical, and there is some truth there. The main differences between KN95 and N95 masks don’t really impact their ability to protect from COVID-19. Regardless, they do bear mentioning.
A KN95 mask needs to go through a general mask fit test to get certified. To do this, the need to have 8% or lower leakage from the mask. This is a major difference people don’t get between surgical and respirator masks. Having a proper fit and seal is important because otherwise, particles can travel around the edge of your mask and get behind it to enter your mouth or nose. To give you an idea, the average surgical masks have a leakage rating of around 37%. N95 masks don’t have a fit test standard like this, but fit tests are still done. The difference is that fit tests are normally administered by organizations (a hospital, a business) when they get their private shipments, rather than being done by the manufacturer as they are made.
 The other major difference between the two is breathability. This is important for comfort, and that shouldn’t be ignored. People who can’t wear a mask comfortably are more likely to fidget or mess with it, which can break the seal and potentially leave you exposed. This is the one area where N95 masks have a noted advantage over KN95, requiring a smaller standard of inhalation and exhalation resistance. Note that the differences aren’t huge between the two, but they are going to create a difference in terms of how you can inhale and exhale in those masks. People who are going to be doing physically intensive work in their masks, like first responders, should be mindful of this.
The other major difference between the two is breathability. This is important for comfort, and that shouldn’t be ignored. People who can’t wear a mask comfortably are more likely to fidget or mess with it, which can break the seal and potentially leave you exposed. This is the one area where N95 masks have a noted advantage over KN95, requiring a smaller standard of inhalation and exhalation resistance. Note that the differences aren’t huge between the two, but they are going to create a difference in terms of how you can inhale and exhale in those masks. People who are going to be doing physically intensive work in their masks, like first responders, should be mindful of this.
So, how does the EUA factor into this? Normally, N95s would be the only option available to places like hospitals or other essential settings. However, with demand being so high, that restriction would lead to countless mask shortages. With the main standard differences being unrelated to protection, the EUA helps cut through red tape and makes sure that more people get the protection they need.
In April, the FDA issued an EUA allowing for usage of respirators not approved by NIOSH, like the KN95. The reasoning here is that, as we covered, the devices were evaluated by measures similar to NIOSH, and were expected to provide an adequate measure of protection. In essence, what this means is that the government recognizes that slightly different masks are better than having no respirator masks at all, and the EUA allows for a temporary loosening of restrictions to have more protection.
Note that the EUA isn’t a blanket allowance for all masks labeled KN95. It only includes masks made in certain Chinese factories, so you need to do your research when you search.
Also, it bears mentioning that the terms of a EUA aren’t set in stone. The government and CDC work together to make sure that the masks they are approving still meet those performance standards. They did just that in May in order to remove some that didn’t meet the criteria. They have also implemented random testing on respirator shipments from China to ensure quality standards are met. So, in a nutshell, what does the EUA mean about your mask purchases?
- Normally, only N95 would be approved
- There aren’t enough N95 masks to meet public needs
- KN95 masks have similar protection, but different standards in other areas
- The EUA allows for KN95 masks from certain factories to be used to address the need
- EUAs can be revised as needed to either tighten restrictions or react to other developments
 Your EUA KN95 Mask Options
Your EUA KN95 Mask Options
So, where should you start in terms of purchasing your EUA KN95 masks? We have you covered with the KN95 Respirator Face Masks – N95 Alternative (EUA) Factory. As mentioned before, in terms of protective functionality, these strong, breathable, full mesh nylon masks are similar to the lauded N95 mask and have both a KN95 FDA Certificate and KN95 CE Certificate.
In terms of wearing on a day-to-day basis, this mask is designed to keep you comfortable and protected, key for people who need to wear a mask as part of long work shifts. The tip-shaped hanging ear design reduces slippage, while the silicone adjustable nose clip allows for the all-important face seal, as well as keeping glasses from fogging up.
At iPromo, we source our EUA KN95 masks from this list of factories. Have all the protection you need for your team, as well as additional gear to build up your workplace PPE stockpile. Compared to other KN95 options you may buy, this gives you a little more consumer confidence in the product, knowing that the manufacturing facility is monitored by the FDA.
As a final point, scarcity or time concerns may mean that you look to a variety of different vendors in order to source your KN95 masks. However, it’s extremely important that you test out the authenticity of any masks that you purchase, especially if it’s a EUA product. While this measure opens up a lot of potential PPE for usage in essential settings, malicious actors may take advantage of the measure to try and sell inferior products. The good news is there are options to test out KN95 mask function, here are the three main tests.
Lighter mask testing: For this test, you hold a lighter, candle, or similar small flame six inches from your mouth, then try to blow it out, like a birthday cake. If you have a proper mask, no matter how hard you try, this shouldn’t work because of the nylon in a proper mask.
Water testing: To administer this test, hold your mask by its elastic ear bands and fill it with water. All proper KN95 masks are waterproof, so there should be no leaks. In practice, this property protects you against any biological fluids, like droplets from sneezing or coughing.
Sweetener testing: This test requires you to put on the mask, then put a packet of sweetener either on a spoon or flat surface. You want to smell it with the mask, on, then the mask off. If you have a quality mask, the filtration should mean you only get a small scent with on versus when it’s off.
Use any one of these methods with our masks and you’ll have a key trust marker of their quality. These methods are also useful to test any future KN95 masks that you buy.
Having PPE that you can trust, KN95 masks included, is essential to make your clients, customers, students, parents, or any other figures confident in your organization’s ability to manage COVID-19. To do that, you need a provider that you can trust. At iPromo, our goal is to fill that need with all the essential PPE you need. Not only do we make sure we have KN95 masks are made in factories approved with the current EUA, but we also have 20 years of international sourcing to ensure you get what you need when you need it.